Translate this page into:
Impact of platelet-rich fibrin on pain after mandibular third molar extraction: A systematic review and meta-analysis

-
Received: ,
Accepted: ,
How to cite this article: Raja K, Devi K. Impact of platelet-rich fibrin on pain after mandibular third molar extraction: A systematic review and meta-analysis. J Global Oral Health 2023;6:43-9.
Abstract
Platelet rich fibrin (PRF) is an autologous biomaterial and has many clinical applications. It is produced without the addition of anticoagulant and with no gelling agent, through the immediate centrifugation of blood after collection. The effect of PRF on post-surgical pain is unclear. Thus the aim of this systematic review was to assess whether PRF was effective in controlling pain after the extraction of bilateral mandibular third molars. A comprehensive literature search was performed for articles published from January 2000 to January 2021 in the PubMed, PubMed Central, Directory of Open Access Journals (DOAJ), Campbell systematic review, Scopus, Cochrane review, Embase and Google scholar. Randomized clinical trials with PRF used as one of the comparison groups PRF after extraction and articles assessing pain as an outcomes measure were included for the systematic review. Results: A total of 160 records were found by preliminary screening through database search and 30 records were retrieved by additional sources, of which eleven were selected for qualitative analysis and four for quantitative analysis. A meta-analysis was performed only for pain, due to the considerable heterogeneity among studies for the other outcome variables. The overall quality of evidence was low for all outcomes of included studies. In these studies pain was assessed using a visual analog scale ranging from 0 (no pain) to 10 (most severe pain) point scale. Post-operative pain on day 1, 3 and 7 showed statistically significant difference between the PRF and non-PRF groups. In conclusion PRF administered after third molar extraction significantly reduced pain. But, further randomized controlled trials with larger cases and well-designed models are essential to validate the current findings.
Keywords
Mandibular third molar
Pain
Platelet-rich fibrin
INTRODUCTION
Third molar (M3) extraction is one of the most common operations in oral and maxillofacial surgery. The literature on M3 removal consequences is vast.[1]
After the third molar tooth is removed, pain, edema, inflammation, alveolar osteitis (dry socket), and bleeding are all possible post-operative consequences. To prevent post-operative problems, oral and maxillofacial surgeons are continuously working to enhance their surgical technique.[2]
Inflammatory pain is caused by tissue damage and inflammation, neuropathic pain is caused by damage to the central nervous system, and functional pain is caused by changes in the normal function of the nervous system.[3] Pain is a key issue that might discourage patients from obtaining dental treatment in clinical settings.[4,5]
The extraction of an impacted molar may cause severe agony to the patient. It begins when the anesthetic wears off and peaks on the first day following surgery. If a dry socket or infection arises, inflammation can make it more difficult to alleviate post-operative discomfort.[6,7]
This is classified as being of short to moderate duration, with the peak severity occurring during the first 12 h after surgery.[8,9] As a result, oral and maxillofacial surgeons are continuously refining their surgical techniques to reduce post-operative problems.
Using a centrifuge to produce a platelet-rich fibrin (PRF) clot from autogenous whole blood, Dr. Joseph Choukroun of France was the first to discuss the use of PRF in oral and maxillofacial surgery.[10] To accelerate socket repair and reduce post-operative discomfort, he suggested utilizing a platelet concentrate autogenous substance (PRF) in tooth extraction sites.[11] PRF is part of a new class of platelet concentrates that are simple to prepare and do not need biological blood processing. PRF is a kind of autologous tissue that may be employed in a number of therapeutic applications. It is created without anticoagulants or gelling chemicals, and it is made by centrifuging blood just after it has been processed.[11] One of the main benefits of PRF over first-generation platelet concentrate platelet-rich plasma (PRP) is the complex and resilient structure of the fibrin matrix, which stores a considerable number of cytokines inside its structure and releases them slowly during healing (PRP).[12] The fibrin matrix’s particular design, in combination with the leukocyte material it contains, may have a healing impact, lowering surgery-related morbidity. PRF had demonstrated good outcomes in the treatment of bony deformities, maxillary sinus augmentation, dental implant surgery, periodontal tissue engineering, post-extraction socket repair, and third molar surgery.[13] Some researchers have discovered that PRF can help reduce discomfort following mandibular third molar surgery in particular.[14]
The real impact of PRF on post-operative problems, however, is unclear. The goal of this systematic study was to see if PRF was effective in controlling pain after bilateral mandibular third molar extractions. The hypothesis for this review was that applying PRF locally to the extraction site of the lower third molars would minimize post-operative pain.
MATERIALS AND METHODS
The parameters for this systematic review were based on the 2009 Preferred Reporting Items for Systematic Reviews and Meta-Analysis declaration.[15] A detailed protocol was developed prior and was registered in the PROSPERO database (Registration number CRD42020187688).
Multiple electronic databases such as PubMed, PubMed Central, Directory of Open Access Journals (DOAJ), Campbell systematic review, Scopus, Cochrane review, Embase, and Google Scholar were used to conduct a conventional literature search. This review covered all English-language articles that were published between January 2000 and January 2021.
The articles with following criteria were included for the review
Randomized and controlled trials assessing bilateral mandibular third molars impaction requiring surgical extraction
Randomized and controlled trials with at least two comparison groups: One group receiving PRF after extraction and the other group receiving control treatment without PRF after extraction
Articles published in the English language
Articles assessing pain as an outcomes measure.
The exclusion criteria were as follows
Randomized and controlled trials with PRF placed at the extraction sites of maxillary third molars
Randomized and controlled trials without comparison group
Letter to editor, case report, personnel proceedings, personal communications, and any types of reviews were excluded from the study
Systematic reviews and meta-analysis
Studies published in language other than English.
Search strategy
PubMed, PubMed Central, EMBASE, Google Scholar, DOAJ, Scopus, Campbell systematic review, and Cochrane were used to conduct a complete electronic search. The search was conducted using medical subject heading phrases as well as free text words. To identify the relevant papers, researchers used keywords such as platelet-rich plasma, PRF, pain, tooth extraction, mandibular third molar, impaction, and postoperative problems. This evaluation covered studies that were published up through January 2021. The reference lists of the eligible paper as well as review articles were manually searched.
The search for suitable research was conducted until January 30, 2021. During the search process, if any relevant publications were discovered without full text, the respective authors were contacted through email to obtain the article. Duplicate articles were initially eliminated. The study’s titles and abstracts were then evaluated separately by two writers using the eligibility criteria.
Finally, the inclusion and exclusion criteria were applied to the entire text of the articles that were initially included in the study. If the two author’s options differed during the process, they came to an agreement through conversation. The inter examiner reliability was measured using Cohen’s kappa.
Extraction of data
The first author used a data extraction form to obtain data from the included studies, which was then confirmed by consensus with the second author. The following information was gathered from each article: For PRF sample collection, volume of blood drawn and centrifugation parameters such as speed and time were gathered from included studies [Table 1]. For description of included studies: The first author’s surname, the year, the article was published, the location of the study, the mean age or range, the number of participants in the test and control groups, the number of follow-up visits, and the outcome measured. Two authors independently derived these results. Any disagreements were handled between them, and in the end, they came to an agreement [Table 2].
| Author, Year | Volume of blood drawn, in ml | Centrifugation parameters speed and time |
|---|---|---|
| Sybil et al.,[17] 2020 | 10 ml | 3000 rpm for 10 min |
| Bhujbal et al.,[18] 2020 | 6 ml | 3000 rpm for 10 min |
| Surekha et al.,[19] 2019 | 10 ml | 3000 rpm for 10 min |
| Kapse et al.,[20] 2019 | 10 ml | 2700 rpm for 12 min |
| Singha et al.,[21] 2019 | 10 ml | 3000 rpm for 15 min |
| Dar et al.,[22] 2018 | 10 ml | 3000 rpm for 12 min |
| Unakalkar et al.,[23] 2018 | NR | NR |
| Gulsen et al,[24] 2017 | 10 ml | 3000 rpm for 10 min |
| Asutay et al.,[25] 2017 | 12 ml | 2700 rpm for 12 min |
| Kumar et al.,[26] 2015 | 5 ml | 3000 rpm for 10 min |
| Ozgul et al.,[27] 2015 | 10 ml | 3000 rpm for 10 min |
NR: Not reported, PRF: Platelet-rich fibrin
| Author, Year | Place of study | Mean age, (Range) | Number of participants | Follow-up visits (Post-operative visits) |
Outcome assessed | |
|---|---|---|---|---|---|---|
| Test | Control | |||||
| Sybil et al.,[17] 2020 | India | 32.3 (18–55) | 25 | 25 | Pain, tenderness, and edema – 1st, 3rd day, 1 week, and 1 month. Sensitivity – 1 week, 1, 3, and 6 months. Area-specific SBI and Plaque Index – 1 week and 1, 3, and 6 months. Area-specific CAL, probing depth, and radiographic bone height – 3 and 6 months. |
Pain, edema, tenderness, sensitivity, SBI, Plaque Index, CAL, probing depth, and bone height |
| Bhujbal et al.,[18] 2020 |
22 | 20 | 20 | Soft-tissue healing, pain, and swelling – 1st day, 3rd day, and 7th day. Radiological bone density – 3rd and 6th month. |
Soft-tissue healing, pain, and swelling | |
| Surekha et al.,[19] 2019 |
15 | 15 | Pain, facial swelling, soft-tissue healing, – 1st, 3rd, and 7th day. Bone density – 1st and 3rd month. |
Pain, facial swelling, soft-tissue healing, and bone density | ||
| Kapse et al.,[20] 2019 |
India | 25.47±0.90 years 18–40 |
30 | 30 | Pain and swelling – 1, 3, 7, and 14 day. Bone healing – 8th and 16th post-operative week. |
Pain, swelling and bone healing. |
| Singha et al.,[21] 2019 |
Madhya Pradesh, India. | 18–30 years | 100 | 100 | Pain, Facial measurements, mouth opening, dry socket, and infection – 1st, 3rd, and 7th day. Wound dehiscence – 3rd and 7th day Soft tissue healing – 7th day |
Pain, facial measurements, mouth opening, dry socket, infection and wound dehiscence, and soft-tissue healing. |
| Dar et al.,[22] 2018 | Srinagar, Jammu and Kashmir, India. | 18–50 | 60 | 60 | Pain – 1st, 3rd, 7th, and 14th day. Swelling – 1st, 3rd, 7th, and 14th day Bone healing – 4th and 12th week |
Pain, swelling, periodontal health, and bone healing. |
| Unakalkar et al.,[23] 2018 |
India | 18–40 | 25 | 25 | Pain – 1st, 3rd, 7th, and 14th day. Trismus – 1st, 3rd, 7th, and 14th day. Swelling – 1st, 3rd, 7th, and 14th day. Periodontal health– 8th and 16th week. Bone healing – 8th and 16th week. |
Pain, swelling, trismus, periodontal health, and bone healing. |
| Gulsen et al.,[24] 2017 |
Turkey | 17–27 | 30 | 30 | Edema – 2nd and 7th day. Pain – 6 h, 12 h. 1st, 2nd, 3rd, and 7th day. |
Edema and pain |
| Asutay et al.,[25] 2017 |
Turkey | 20.32 (18–29 years) |
30 | 30 | Pain – 6 h, 12 h, and 1, 2, 3, 4, 5, 6, and 7 days after surgery. Trismus and mouth opening 2nd and 7th Post-operative days. |
Pain, trismus, and mouth opening |
| Kumar et al.,[26] 2015 |
India | 19–35 years | 16 | 15 | Pain, swelling, and mouth opening – 1st, 1, and 3 month. Periodontal pocket depth and bone formation – 1 and 3 month. |
Pain, swelling, maximum mouth opening, periodontal pocket depth and bone formation. |
| Ozgul et al.,[27] 2015 |
Turkey | 18–28 years | 56 | 56 | Pain and facial swelling – 1, 3, and 7 days. | Pain and facial swelling |
CAL: Clinical attachment level, SBI: Sulcus bleeding index
Quality assessment
The studies included in the review were analyzed using the Cochrane collaboration method driven by the Cochrane Handbook for Systematic Intervention Reviews (RevMan, version 5.3 The Cochrane Collaboration, Copenhagen, Denmark).[16] The risk of bias in the studies that were included was rated as “low,” “unclear,” or “high.” Method of sequence creation, allocation sequence concealment, blinding of care providers and participants, blinding of outcome assessors, inadequate outcome data, selective outcome reporting, and other forms of bias were all examined to determine the risk of bias in each research. Any discrepancies between the writers were worked out through communication. Review manager version 5.3 software was used for performing the analysis.
RESULTS
A first database search yielded 160 entries and further sources yielded 30 more records. Out of the 190 papers, 111 were evaluated for eligibility, with 96 articles failing to fulfill the inclusion criteria and just 11 being included in the review. The flow diagram for the selection of articles included in this study is shown in [Figure 1]. [Table 1] shows method of PRF preparation for all included studies.[17-27]

- Search strategy.
The overall quality of evidence was low for all outcomes. The risk of bias was evaluated as unclear in most studies, except for selective reporting, because of lack of detail in the reports. The assessments of risk of bias for included studies are given in [Figures 2 and 3]. Only two studies (5/14) described the random sequence generation and allocation concealment while detailed information was deficient in the other nine trials. None of the trials (0/11) described blinding of participants and personnel clearly being judged to be “high” in this domain. The majority of trials (5/11) provided no details related to blinding of outcome assessment and for five (5/11) judged to be “unclear” due to lack of details. Only five trials (5/11) showed low risk of bias for incomplete outcome data according to the reports of the drop-outs or intention-to-treat analysis. Majority of the studies (11/12) had low risk of bias for selective reporting and other bias.
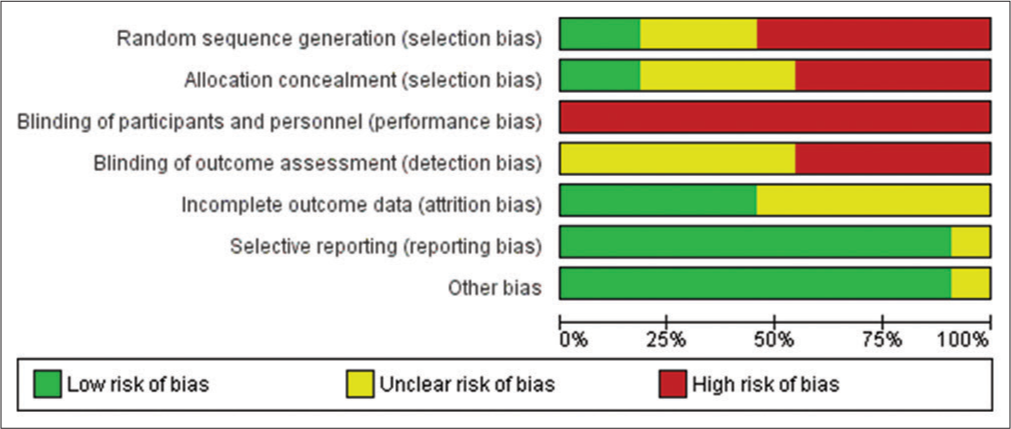
- Review author’s judgment’s about each risk of bias item presented as percentages across all included studies.

- Risk of bias overview for evaluating the judgments of the author on each risk of bias items for each study included. Green stands for a low bias risk, yellow reflects an unknown bias risk, and red suggests a high bias risk.
A total of 260 cases from four RCTs were included in the meta-analysis of pain. In these studies, pain was assessed using a visual analog scale ranging from 0 (no pain) to 10 (most severe pain) point scale. For day-1 post-operative pain, there was statistically significant differences (Weighted Mean Difference [WMD], −1.05; 95% CI, −1.33 to −0.77; P = 0.00001) between the PRF and non-PRF groups [Figure 4]. Similarly, significant difference was observed for day-3 post-operative pain between the PRF and non-PRF groups (WMD, −1.00; 95% CI, −1.28 to −0.72; P = 0.00001) [Figure 5]. Day-7 also showed significant difference between the PRF and non-PRF groups (WMD, −0.67; 95% CI, −1.01 to −0.33; P = 0.0001) [Figure 6].

- Forest plot and meta-analysis comparing day-1 post-operative pain for 10 point VAS scale. SD: Standard deviation

- Forest plot and meta-analysis comparing day-3 post-operative pain for 10 point VAS scale. SD: Standard deviation

- Forest plot and meta-analysis comparing day-7 post-operative pain for 10 point VAS scale. SD: Standard deviation
DISCUSSION
Slow polymerization occurs throughout preparation, resulting in a fibrous protein network that promotes cell motility and proliferation, similar to that observed in natural cells. PRF is widely utilized as a direct graft or in combination with allograft or xenograft in the oral and maxillofacial areas.[28] In addition, without the use of a flap, PRF clots are utilized to repair acute sinus perforations.[29] Socket retention, infrabony flaws, and periodontal issues are among the other indications for PRF usage.[11] The present meta-analysis looked at the influence of PRF on a mandibular third molar in 260 cases from four studies, with the goal of evaluating the effect of PRF on post-operative pain following mandibular third molar surgery.
This might aid the oral and maxillofacial surgeon in giving a better post-operative outcome for his/her patients. Only pain was used as an outcome measure in this meta-analysis. The meta-analysis for these additional factors was not possible due to methodological heterogeneity across the included studies.
The findings of this meta-analysis revealed that when PRF was used, pain was reduced on the 1st, 3rd, and 7th days following surgery, with a statistically significant difference between the PRF and non-PRF groups. These findings point to PRF having a therapeutically positive effect on post-operative pain, as well as confirming the notion that applying PRF following lower third molar extraction reduces post-operative discomfort. Although these findings are encouraging, due to the significant possibility of bias in the studies included, care is advised to avoid misunderstanding of the real data.
Our findings matched those of Xiang et al.,[30] who found that applying PRF locally during the extraction of the lower third molar considerably reduced discomfort on the 3rd post-operative day. Canellas et al.[13] and Al-Hamed et al.[14] both show that PRF has a clinically positive impact on post-operative pain.
A meta-analysis could be performed only for pain. The lack of a meta-analysis for various additional variables, such as edema, trismus, soft-tissue healing, and bone density, was due to the high methodological heterogeneity across the included studies. The randomized procedure and allocation concealment are the most essential components of a clinical trial in terms of risk of bias analysis. Only two of the 11 publications included in this study explained the process for generating the allocation sequence in sufficient depth, as well as the strategy for concealing the allocation sequence. Inadequate allocation concealment can lead to incorrect treatment effect estimates. The current meta-analysis has the following drawbacks: To begin with, the number of research included was limited, potentially reducing statistical power. Second, only studies that used the same measuring scale were considered. Third, only papers published in English were considered. Finally, the length of follow-up in the four trials differed significantly, making it difficult to determine the long-term clinical effects of PRF on the mandibular third molar.
CONCLUSION
Despite the limitations, this study shows that PRF given after third molar extraction considerably reduces discomfort. To corroborate the current findings, more study with larger instances and well-designed models is required.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Conflicts of interest
There are no conflicts of interest.
Financial support and sponsorship
Nil.
References
- Types, frequencies, and risk factors for complications after third molar extraction. J Oral Maxillofac Surg. 2003;61:1379-89.
- [CrossRef] [PubMed] [Google Scholar]
- Design of a scale for measuring post-surgical complications in third molar surgery. Int J Oral Maxillofac Surg. 2014;43:1008-14.
- [CrossRef] [PubMed] [Google Scholar]
- Prognostic factors affecting the duration of disability after third molar removal. J Oral Maxillofac Surg. 2011;69:1272-7.
- [CrossRef] [PubMed] [Google Scholar]
- Dental pessimism: Negative cognitions in fearful dental patients. Behav Res Ther. 1984;22:553-6.
- [CrossRef] [PubMed] [Google Scholar]
- Modern concepts in understanding and management of the “dry socket” syndrome: Comprehensive review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;107:30-5.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical concepts of dry socket. J Oral Maxillofac Surg. 2010;68:1922-32.
- [CrossRef] [PubMed] [Google Scholar]
- Morbidity of third molar extraction in patients between 12 and 18 years of age. Med Oral Patol Oral Cir Bucal. 2005;10:422-31.
- [Google Scholar]
- Efficacy of nimesulide versus meloxicam in the control of pain, swelling and trismus following extraction of impacted lower third molar. Int J Oral Maxillofac Surg. 2010;39:580-4.
- [CrossRef] [PubMed] [Google Scholar]
- Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part I: Technological concepts and evolution. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101:e37-44.
- [CrossRef] [PubMed] [Google Scholar]
- Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part IV: Clinical effects on tissue healing. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101:e56-60.
- [CrossRef] [PubMed] [Google Scholar]
- Classification of platelet concentrates: From pure platelet-rich plasma (P-PRP) to leucocyte-and platelet-rich fibrin (L-PRF) Trends Biotechnol. 2009;27:158-67.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of postoperative complications after mandibular third molar surgery with the use of platelet-rich fibrin: A systematic review and meta-analysis. Int J Oral Maxillofac Surg. 2017;46:1138-46.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical effects of platelet-rich fibrin (PRF) following surgical extraction of lower third molar. Saudi J Dent Res. 2017;8:19-25.
- [CrossRef] [Google Scholar]
- Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann Intern Med. 2009;151:264-9. w64
- [CrossRef] [PubMed] [Google Scholar]
- Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 United Kingdom: The Cochrane Collaboration; 2011.
- [Google Scholar]
- Platelet-rich fibrin for hard-and soft-tissue healing in mandibular third molar extraction socket. Ann Maxillofac Surg. 2020;10:102-7.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of plateletrich fibrin and plateletrich plasma in impacted mandibular third molar extraction socket healing and bone regeneration: A splitmouth comparative study. Eur J Gen Dent. 2020;9:96102.
- [CrossRef] [Google Scholar]
- Efficacy of autologous platelet-rich fibrin in osseous regeneration after mandibular third molar surgery: A comparative study. J NTR Univ Health Sci. 2019;8:37-41.
- [Google Scholar]
- Autologous platelet rich fibrin: Can it secure a better healing? Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;127:8-18.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of treatment outcome after impacted mandibular third molar surgery with and without autologous platelet concentrates. SRM J Res Dent Sci. 2019;10:72-7.
- [CrossRef] [Google Scholar]
- Healing potential of platelet rich fibrin in impacted mandibular third molar extraction sockets. Ann Maxillofac Surg. 2018;8:206-13.
- [CrossRef] [PubMed] [Google Scholar]
- Comparision of the efficacy of platelet-rich fibrin with plateletrich plasma in third molar extraction socket-a prospective clinical study. Int J Oral Care Res. 2018;6:S44-9.
- [Google Scholar]
- Effect of platelet rich fibrin on edema and pain following third molar surgery: A split mouth control study. BMC Oral Health. 2017;17:79.
- [CrossRef] [PubMed] [Google Scholar]
- An evaluation of effects of platelet-rich-fibrin on postoperative morbidities after lower third molar surgery. Niger J Clin Pract. 2017;20:1531-6.
- [Google Scholar]
- Evaluation of treatment outcome after impacted mandibular third molar surgery with the use of autologous platelet-rich fibrin: A randomized controlled clinical study. J Oral Maxillofac Surg. 2015;73:1042-9.
- [CrossRef] [PubMed] [Google Scholar]
- Efficacy of platelet rich fibrin in the reduction of the pain and swelling after impacted third molar surgery: Randomized multicenter split-mouth clinical trial. Head Face Med. 2015;11:37.
- [CrossRef] [PubMed] [Google Scholar]
- Comparative radiologic analyses of newly formed bone after maxillary sinus augmentation with and without bone grafting. J Oral Maxillofac Surg. 2013;71:1520-30.
- [CrossRef] [PubMed] [Google Scholar]
- Flap-free treatment of an oroantral communication with platelet-rich fibrin. Br J Oral Maxillofac Surg. 2016;54:702-3.
- [CrossRef] [PubMed] [Google Scholar]
- Impact of platelet-rich fibrin on mandibular third molar surgery recovery: A systematic review and meta-analysis. BMC Oral Health. 2019;19:163.
- [CrossRef] [PubMed] [Google Scholar]






